 Medium = substitution is possible but there may be an economic and/or performance impact High = substitution not possible or very difficult. The availability of suitable substitutes for a given commodity. A higher recycling rate may reduce risk to supply. The percentage of a commodity which is recycled. The number of atoms of the element per 1 million atoms of the Earth’s crust. This is calculated by combining the scores for crustal abundance, reserve distribution, production concentration, substitutability, recycling rate and political stability scores. The Chemical Abstracts Service registry number is a unique identifier of a particular chemical, designed to prevent confusion arising from different languages and naming systems.ĭata for this section been provided by the British Geological Survey.Īn integrated supply risk index from 1 (very low risk) to 10 (very high risk). Where more than one isotope exists, the value given is the abundance weighted average.Ītoms of the same element with different numbers of neutrons. This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. 
Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. Learn about the Floridan aquifer system next.A vertical column in the periodic table. If you live in Florida, there’s a good chance that the water you drink every day comes from under the ground - the hidden part of the water cycle. We put pipes in the holes and pump up the good water. Sometimes groundwater naturally comes to the surface in springs, but most people get it by digging holes called wells. Water in an aquifer is usually cleaner than surface water, especially if it’s deep or has been underground a long time. This groundwater becomes part of an aquifer, which is an underground water supply. It’s water that’s hidden below our feet. When it rains, some of that water will seep into the ground. This hidden place is where most people in Florida get their drinking water. But there’s also another way to get fresh, clean water. When we use surface water for drinking, we treat it first to make it safe - with chemicals or filters or even ultraviolet light. 
Why not? Because many surface waters contain pollutants that can make you sick if you drink them. 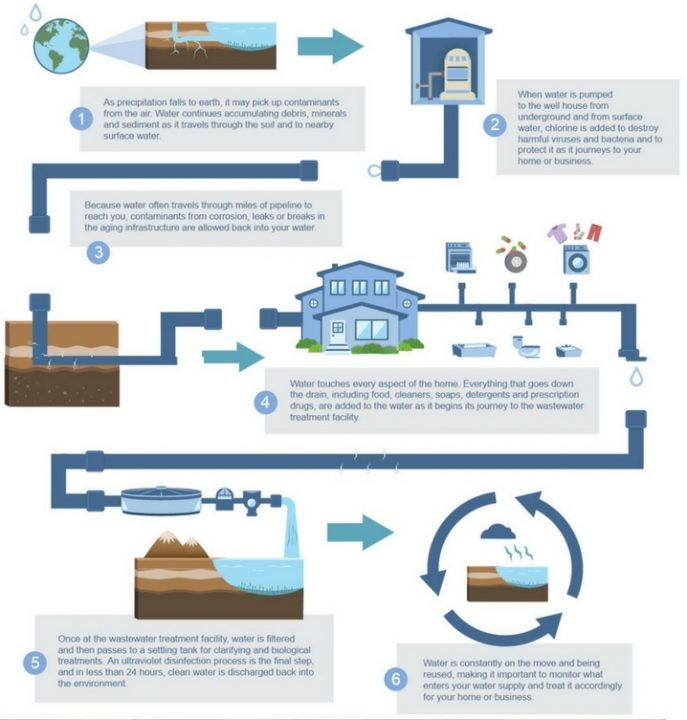
But would you drink from the ditch near your house? No! So where do you get your drinking water from? In Florida, lakes, rivers and swamps all contain water that isn’t frozen or too salty. 
Not just any fresh water though. Did you know most of the world’s fresh water is frozen solid? Nearly 80% is locked up in ice sheets and glaciers. The ocean? That would be a good guess because the oceans contain most of earth’s water, about 97%! But would you drink a glass of seawater? Do you know where your drinking water comes from? You might have poured it from a tap or bought it at the store, but it had to come from somewhere before that. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
